The immune system is the body’s army against bacteria, viruses or any foreign pathogen that can cause disease. This innate defense system also protects the body against irregularities, such as cancer cells, by marking them as “dangerous” and targeted for destruction.
Inducing cell death has been a significant area of interest for cancer therapy, with one notable pathway of cell death gaining traction since its discovery a decade ago: ferroptosis. New research published in Nature reveals that ferroptosis is doing more than marking cells for death – it’s also sending signals that influence the immune response.

Dr. Valerian Kagan
“Ferroptosis is an exploding field,” said study co-author Dr. Valerian E. Kagan, professor in the University of Pittsburgh School of Public Health’s Department of Environmental and Occupational Health. “Understanding exactly how it works in not only destroying tumor cells, but also in helping immune cells function correctly to fight cancer will show doctors when to induce ferroptosis and when to inhibit it.”
Ferroptosis is a specific type of programmed cell death that is dependent on iron (“ferro” meaning iron in Latin), as this element participates in electron-transfer reactions, also known as redox reactions, that can damage cells.
Ferroptosis-inducing drugs are effective at killing cancer cells in a dish. These drugs also greatly reduce tumors in immunodeficient mice with cancer. However, mice that have working immune systems do not respond as well to ferroptosis-inducing drugs.
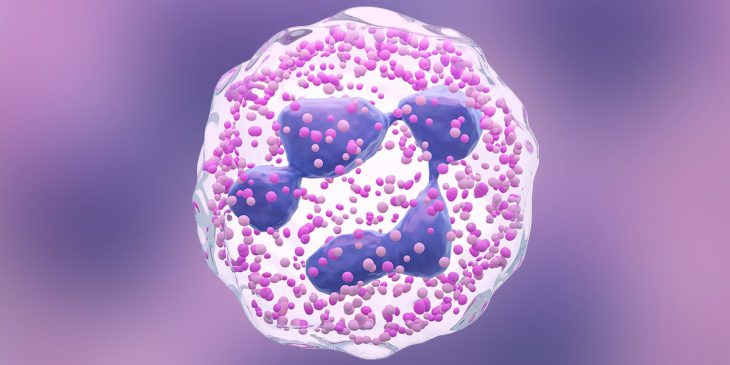
Image of a neutrophil, which is one type of white blood cell.
The newly published, National Institutes of Health-funded study investigates this discrepancy.
Kagan and Dr. Dmitry I. Gabrilovich, chief scientist of cancer immunology at AstraZeneca, found that pathologically activated neutrophils, unlike normal neutrophils that inhibit cancer, actually suppress anti-tumor immune response and are associated with poor clinical outcomes in cancer. In the tumor microenvironment, ferroptosis spontaneously kills pathologically activated neutrophils, suggesting that inducing ferroptosis could be a therapeutic target in cancer therapy. However, the new study found that ferroptosis also causes the release of immunosuppressive molecules from these neutrophils, which inhibit another important class of cancer-fighting cells called T cells. As such, the researchers found that inducing ferroptosis actually promoted tumor growth in mice with functional immune systems.
“The word ferroptosis initially meant only death – a ferroptotic type of death,” said Kagan. “Now this has to be revised because we’ve shown ferroptosis is a much broader response of cells to dysregulation of redox reactions.”
Future research will need to characterize the cascading signals set off by the redox reactions that instruct cells to undertake various actions, Kagan said. By understanding this process, doctors may someday be able to incorporate a precision medicine approach to using ferroptosis to treat cancer, inhibiting or inducing it depending on whether the patient’s immune system is functional.
Lisa Hong is a Ph.D. student in the University of Pittsburgh School of Medicine’s Molecular Pharmacology Program. She is participating in the UPMC Science Writing Mentorship Program.