Today, the only cure for end-stage liver disease is an organ transplant – an expensive treatment, the demand for which outweighs the supply. Nearly 12,000 people in the U.S. are waiting for a liver right now.
But what if the same technology as the Moderna and Pfizer COVID-19 vaccines could prompt a diseased liver to cure itself, eliminating the need for a transplant?

Dr. Alejandro Soto-Gutierrez
University of Pittsburgh School of Medicine and UPMC physician-scientists have done just that in rats – and now they’ve set their sights on humans.
“What we’ve discovered could transform organ transplantation,” said Dr. Alejandro Soto-Gutierrez, associate professor of pathology at Pitt. “We’re talking about, essentially, a vaccine that might bring patients back from terminal liver failure, no surgery needed.”
Soto-Gutierrez partnered with UPMC transplant surgeon Dr. Ira Fox – both members of the Pittsburgh Liver Research Center and the McGowan Institute for Regenerative Medicine – to explore gene expression and the possibility of “reprogramming” failing liver cells.

Dr. Ira Fox
Over the past several years, they zeroed in on a transcription factor called P1-HNF4 alpha, which acts as a main control panel, regulating much of the gene expression in liver cells. The researchers found that when P1-HNF4 alpha was turned off in rats, they’d experience liver failure, but re-expression of the transcription factor restored liver function.
Then, the team corroborated that P1-HNF4 alpha was the best target for end-stage liver disease in humans by analyzing nearly 100 failing livers donated by UPMC transplant patients.
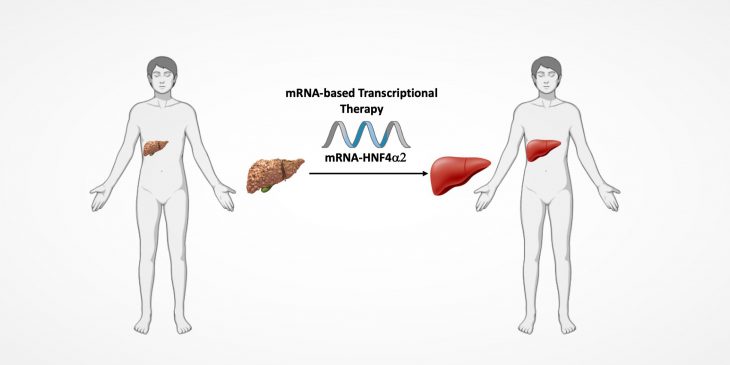
In their most recent study – published this month in Hepatology Communications – the researchers describe the results of treating cells from failing human livers in the lab with a messenger RNA (mRNA) vaccine that delivered instructions to turn the transcription factor back on. The treatment worked.
“There are a lot of genes in liver cells that depend on transcription factors to perform all the essential functions of the liver,” said Soto-Gutierrez. “When supplemented with our mRNA treatment, all those functions came back in a matter of about 24 hours. Basically, what we believe we have is a new way to treat chronic diseases.”
The clinical research team is meeting with investors and pharmaceutical companies to develop partnerships to translate their findings into clinical trials. If all goes well, Soto-Gutierrez estimates they could begin human clinical trials within two years.