A psychedelic movie of stressed cells under a microscope sent a team of kidney physiologists and biologists from the University of Pittsburgh and Carnegie Mellon University on a journey to solve a mystery: How do cells control their volume?
Their study, published today in Cell, explains how the scientists used a little bit of serendipity to connect the dots on a puzzle first proposed three decades ago.
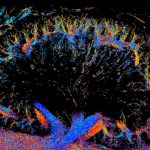
“We were doing live fluorescence imaging experiments that were unrelated to this study, and when we added a salt solution to the cells, the internal cytoplasmic material rapidly turned into a fluorescent lava lamp,” said Dr. Daniel Shiwarski, a postdoctoral research fellow at Carnegie Mellon University, describing how he and his wife, co-lead author Dr. Cary Boyd-Shiwarski, turned a fortuitous bit of experimentation into an unexpected finding.
“I looked at her, and she asked me what was going on, like I was supposed to know,” he said. “And I said, ‘I have no idea, but I think it’s probably something important!’”
When cells are exposed to a sudden outside stressor, such as high levels of salt or sugar, their volume can decrease. In the early 1990s, scientists proposed that cells restore their volume by somehow monitoring their protein concentration, or how “crowded” it was inside the cell. But they didn’t know how the cell sensed overcrowding.
Then, in the early 2000s, a type of enzyme called With-No-Lysine kinases, or “WNKs,” were discovered. For years, scientists suspected that WNK kinases were reversing cell shrinkage, but how they did this was also a mystery.
The new research solves both puzzles, unveiling how WNK kinases activate the “switch” that returns cell volume to equilibrium through a process called phase separation.

From left to right: Dr. Daniel Shiwarski, Dr. Arohan Subramanya and Dr. Cary Boyd-Shiwarski
“The inside of a cell contains cytosol, and generally people think that this cytosol is diffuse, with all kinds of molecules floating around in a perfectly mixed solution,” said senior author Dr. Arohan R. Subramanya, associate professor in the Renal-Electrolyte Division at Pitt’s School of Medicine and staff physician at the VA Pittsburgh Healthcare System. “But there has been this paradigm shift in our thinking of how cytosol works. It’s really like an emulsion with a bunch of little, tiny protein clusters and droplets, and then when a stress such as overcrowding happens, they come together into big droplets that you can often see with a microscope.”
Those liquid-like droplets were the “lava lamp” that Shiwarski and Boyd-Shiwarski were seeing that fateful day when they experimented with adding a salt solution to the cells. They had fluorescently tagged the WNKs, which were diffused throughout the cytosol, causing the whole cell to glow. When salt was added, the WNKs came together, forming large neon green globules that oozed about the cell like the goo in a lava lamp.
The team characterized what they were seeing as phase separation, which is when WNKs condense into droplets along with the molecules that activate the cell’s salt transporters. This step allows the cell to import both ions and water, returning the cell’s volume to its original state within seconds.
Phase separation is an emerging area of interest, but whether or not this process was an important part of cell function has been a controversy.
“There’s a lot of people out there who don’t believe phase separation is physiologically relevant,” explained Boyd-Shiwarski, assistant professor in the Renal-Electrolyte Division at Pitt’s School of Medicine. “They think it’s something that happens in a test tube when you overexpress proteins or occurs as a pathological process but doesn’t really happen in normal healthy cells.”
But over the past six years, the team conducted multiple studies using stressors similar to the fluctuations that occur within the human body to show that phase separation of the WNKs is a functional response to crowding.
Cell volume recovery has implications for human health as well, Subramanya explained: “One of the reasons why we’re so excited is that the next step for us is to take this back into the kidney.”
Other WNKs activate salt transport within kidney tubule cells when potassium levels are low by forming specialized condensates through phase separation, called WNK bodies. Modern Western diets are often low in potassium, so while attempting to regulate cell volume, WNK bodies may contribute to salt-sensitive hypertension.
While the new discovery won’t have immediate clinical applications, the team is excited to take what they’ve learned and explore the connections between WNKs, phase separation and human health. Eventually, their work may lead to better understanding of how to prevent strokes, high blood pressure and potassium balance disorders.
Dr. Julia O’Brien is a postdoctoral fellow in the University of Pittsburgh School of Nursing. She is participating in the UPMC Science Writing Mentorship Program.