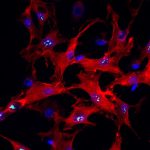
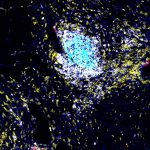
Primary sclerosing cholangitis (PSC) is a long-term autoimmune disease that causes chronic inflammation of the bile ducts (the drainage tubes in the liver) and often affects younger patients. About 15% of patients with PSC go on to develop bile duct cancer, a rare malignancy in the bile ducts. These tumors are already difficult to detect because of their location, and chronic inflammation associated with PSC makes diagnosis even more challenging.
“Treating these patients was my biggest motivation,” said Adam Slivka, M.D., Ph.D., professor of medicine in the Division of Gastroenterology, Hepatology and Nutrition at Pitt. “Seeing young patients with PSC develop bile duct cancer and die underscored the need for a better way to identify cancer earlier in the diagnostic process or detect high-risk genetic changes when standard testing falls short.”
Slivka is part of a group of scientists at UPMC Hillman Cancer Center and the University of Pittsburgh School of Medicine, including the project lead, Aatur Singhi, M.D., Ph.D, associate professor of pathology at Pitt and director of the UPMC Developmental Laboratory, who developed BiliSeq, a molecular test for bile duct samples designed to improve cancer detection, giving physicians a more accurate diagnosis when standard testing falls short.
When evaluated in 327 patients with PSC, pathology alone detected only one in four cancers. By comparison, BiliSeq detected approximately 85%, and when combined with pathology, sensitivity approached 90%.
BiliSeq also demonstrated strong performance in Hispanic patients, a population in which bile duct cancer occurs 50% more often than in other groups. In this group, cancer-related genetic alterations are identified less often, which can make diagnosis more difficult using standard tests. Even in this challenging setting, BiliSeq outperformed standard tissue-based pathology and commonly used blood tests, demonstrating consistent diagnostic accuracy across racial and ethnic groups.
These findings in highrisk patient groups were part of a larger, multiyear study evaluating BiliSeq’s real-world performance. Published today in Gastroenterology, the flagship journal of the American Gastroenterological Association, the study analyzed more than 2,000 patients across the United States and nearly 3,000 bile duct specimens collected over six years. In this broader patient population, BiliSeq nearly doubled cancer detection when combined with pathology, while rarely misclassifying benign disease as malignant — highlighting the test’s strong performance not only in high-risk subgroups, but in bile duct cancer diagnosis overall.
This performance reflects BiliSeq’s ability to detect genetic mutations associated with cancer in bile duct tissue, even when tumor cells are sparse, damaged, or indistinguishable from inflammation under the microscope — a key limitation of traditional pathology that can lead to false-negative‑ results. Together, these findings highlight how molecular testing can help close critical diagnostic gaps for patients who are most likely to be missed by standard approaches.
In addition to improving diagnosis in high-risk patients, the test is already helping guide real-world care decisions, including eligibility for liver transplantation.